Most experts agree that halting climate change—and the global warming, extreme heat events and stronger storms that come with it—will require the removal of carbon dioxide and other greenhouse gasses from the atmosphere. But with humans pumping out an estimated 37 billion metric tons of carbon dioxide annually, current strategies for capturing it seem likely to fall short.
Now, a UCLA research team has proposed a pathway that could help extract billions of metric tons of carbon dioxide from the atmosphere each year. Instead of directly capturing atmospheric carbon dioxide, the technology would extract it from seawater, enabling the seawater to absorb more. Why? Because, per unit volume, seawater holds nearly 150 times more carbon dioxide than air.
The researchers outline their concept, dubbed single-step carbon sequestration and storage, or sCS2, in a paper published today in the journal ACS Sustainable Chemistry & Engineering.
Senior author Gaurav Sant, director of the UCLA Institute for Carbon Management and a Samueli Fellow and professor of civil and environmental engineering and of materials science and engineering at the UCLA Samueli School of Engineering, said:
“To mitigate climate change, we need to remove carbon dioxide from the atmosphere at a level between 10 billion and 20 billion metric tons per year. To fulfill a solution at that scale, we’ve got to draw inspiration from nature.”
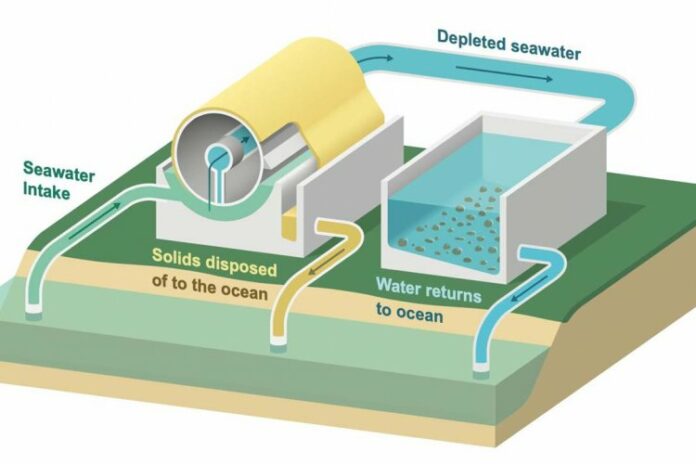
Since the atmosphere and the oceans are in a state of equilibrium, if carbon dioxide were to be extracted from the ocean, carbon dioxide from the atmosphere could then dissolve into it. In this scenario, seawater is like a sponge for carbon dioxide that has already absorbed its full capacity, and the sCS2 process aims to wring it out, allowing the sponge to absorb more carbon dioxide from the atmosphere.

The proposed technology would incorporate a flow reactor—a system that continuously is fed raw materials and yields products. The seawater would flow through a mesh that allows an electrical charge to pass into the water, rendering it alkaline. This kicks off a set of chemical reactions that ultimately combines dissolved carbon dioxide with calcium and magnesium native to seawater, producing limestone and magnesite by a process similar to how seashells form. The seawater that flows out would then be depleted of dissolved carbon dioxide and ready to take up more. A co-product of the reaction, besides minerals, is hydrogen, which is a clean fuel.
sCS2 is meant to durably store carbon dioxide in the form of solid minerals. The first author Erika Callagon La Plante, a former UCLA assistant project scientist, added:
“Durable, safe and permanent storage is the premise of our solution.”
The team carried out detailed analyses of the material and energy inputs and the costs required to realize their concept, as well as what to do with the byproducts. Unsurprisingly, given the enormous magnitude of the carbon challenge, they estimate that it would take nearly 1,800 sCS2 plants to immobilize 10 billion metric tons of carbon dioxide each year, with a cost in the trillions of dollars.
Sant said:
“We should be clear: Managing and mitigating carbon dioxide is foremost an economic challenge. Many of today’s approaches for carbon management either require more clean energy than we can produce or are unaffordable. As such, we need to create solutions that are accessible and that will not impoverish the world. We have tried to use a lens of pragmatism to consider how we may be able to achieve synthetic interventions at an unprecedented scale, while considering the finite energy and financial resources we have.”
Still, the researchers believe that sCS2, even at smaller scales, represents an advance in carbon-capture and storage that should be considered as a potential part of any overall strategy for confronting climate change.